
Chromium also aids in insulin action and glucose breakdown. They are important for brain function and other body processes. It stimulates fatty acid and cholesterol synthesis. How do humans use chromium?Ĭhromium is important in the breakdown of fats and carbohydrates. It’s also used in many dyes and paints, and, when it is applied on top of other metals to make them shiny, it is often just called chrome. Because it has a high melting point, chromium is used to make molds for bricks. Where is chromium used in everyday life?Ĭhromium is an element that has many uses. … Chromium compounds are used as industrial catalysts and pigments (in bright green, yellow, red and orange colours). Chromium plating can be used to give a polished mirror finish to steel. What is chromium used for today?Ĭhromium is used to harden steel, to manufacture stainless steel (named as it won’t rust) and to produce several alloys. Repeated exposure to such objects around the house may cause persistent eczema of the hands. Can chromium cause eczema?įor instance, hexavalent chromium in metal screws and fittings has been shown to cause dermatitis in chromate-sensitive patients. 
What Are the Symptoms of Chromium Deficiency? Consuming too little chromium can result in a range of symptoms, including some that mimic those of diabetes, such as weight loss, impaired glucose tolerance, neuropathy, anxiety, fatigue and muscle weakness, explains Majumdar. What are the symptoms of chromium deficiency? Chromium is represented as Cr and chromate is represented as CrO42. Is chromate and chromium the same thing?Ĭhromium and chromate are different compounds. Zinc chromate, ZnCrO 4, is a chemical compound containing the chromate anion, appearing as odorless yellow powder or yellow-green crystals, but, when used for coatings, pigments are often added. So, the ionic charge on chromate ions is -2. … By adding the oxidation state of chromium and total oxidation state of oxygen, we get -2. It consists of one chromium and four oxygen atoms. When chromate salt dissociates, we get chromate ions. Symptoms may include runny nose, sneezing, coughing, itching and a burning sensation. Can chromium make you itchy?īreathing in high levels of hexavalent chromium can cause irritation to the nose and throat. At the same time, metallic chromium has become one of the most important electroplated metals. It is mainly used in electroplating, tanning, printing, and dyeing, medicine, fuel, catalysts, oxidants, matches, and metal corrosion inhibitors. The Uses of Chromium in the Chemical Industry Animal studies have shown chromium (VI) to cause lung tumors via inhalation exposure. Human studies have clearly established that inhaled chromium (VI) is a human carcinogen, resulting in an increased risk of lung cancer. As nitric acid is added to the potassium chromate solution, the yellow colour turns to orange. The more acidic the solution, the more the equilibrium is shifted to favour the dichromate ion. Yellow chromate ion and orange dichromate ion are in equilibrium with each other in aqueous solution. Why is chromate yellow and dichromate orange? But if you are talking to a plater, ‘dichromate’ often means yellow chromate to them so by contrast ‘chromate’ will mean clear or blue chromate. If you were talking to a chemistry prof, the difference would be that chromate is CrO4 whereas dichromate is Cr2O7. What is the difference between dichromate and chromate? It also has uses in leather tanning, photographic processing, cement, and wood staining. 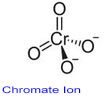
Uses: Potassium dichromate is used for preparing strong cleaning solutions for glassware and for etching materials. Although most people associate ‘chrome’ with the bright, shiny and durable finish of chrome-plated metal products, contact with these rarely give rise to chrome allergy. What are chromium salts found in?Ĭhromium salts are used as an ingredient in the manufacture of many other products such as cement, mortar, leather, paints, and anticorrosives. They can be destroyed by reaction with reducing agents. … They can be made by reaction of chromium(III) oxide and a metal oxide in a basic environment. In these cases, the difference between the ions is in the number of oxygen atoms present, while the overall charge is the same.Chromate is an ion that contains one chromium atom (in its +6 oxidation state) and four oxide atoms. \right)\), there are multiple anions that consist of the same two elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
